J. Mousinga·*, J. Christensena, J. Haugegaarda, A.L. Schirmerb, N.F. Friisb
aFederation of Danish Pig Producers and Slaughterhouses, Axelborg 3, DK-1609 Copenhagen V, Denmark
bNational Veterinary Laboratory, Bülowsvej 27, DK-1790 Copenhagen V, Denmark
Accepted 21 October 1994
ABSTRACT
A total of 796 sows and gilts from 30 Danish sow herds were examined three times at intervals of 6 weeks for serum antibodies to Leprospiru brutisluoa by the microscopic agglutination technique (MAT) test. The prevalence of seroreactors with positive titer values, > 1: 100, at the three successive tests were 2.7%, 2.5% and 2.9%; 4.5% of the animals were positive in at least one of the three tests, and 2.2% showed a greater than two-fold rise in titer between two consecutive samplings. Of the 30 herds, 2 1 (70%) had ever-positive within-herd prevalences in sows and gilts of 4-13%. The risk of a herd having one or more positive sow was positively associated with a herd size of > 141 sows, and distinct regional differences in the prevalence of positive herds were observed. The reproductive performance of the 21 herds with seroreactions was poorer than the performance of the nine herds without positive reactions concerning the variables: ‘days from weaning to last service’ (2.7 days more, P= 0.07), ‘percentage of sows returning to heat’ (4.0 percentage units more, P= 0.03), ‘services per farrowing’ (0.04 more, P = 0.04)) ‘farrowing percentage’ (4.3 percentage units lower, P = 0.06), and ‘stillborn pigs per farrowing’ (0.16 more, P = 0.02).
No association between the MAT serological status of the herd and the incidence of medical treatments of sows and gilts could be found. A high prevalence and low cumulative incidence of seroreactors was demonstrated in first-parity gilts, followed by a low prevalence and cumulative incidence from parity 2 to 3, and a high prevalence and cumulative incidence at the fifth parity.
INTRODUCTION
Infections in swine with Leptospira bratislaua, a parasitic serovar belonging to the Leptospira australis serogroup of the species Leptospira interrogans, are frequent in many swine-producing countries (Bercovich et al., 1983; Sandsted and Engvall, 1985; Egan and Yearsley, 1987; Baker et al., 1988; Ellis, 1992b; Schijnberg et al., 1992). Infections with L. brutisha have been associated mainly with infertility (Ellis, 1992b) but also with abortions. Accordingly, the organism has been demonstrated in the genital tract of sows and boars and from abortions (Ellis and Thiermann, 1986; Ellis et al., 1986a,b; Bolin and Cassells, 1990; Bolin et al., 1991; Ellis, 1992a; Schnberg et al., 1992). L. brutisha is speculated to spread mainly through infected boars. Mice, along with other animals such as dogs and horses, also host L. brutisluou (Ellis, 1992b; Smith et al., 1992).
As microbiological culturing of the organism is very difficult and takes up to 6 months (Bolin et al., 1991; Ellis, 1992b), screening for this infection often is carried out on the basis of serological tests. The standard method used is the microscopic agglutination technique (MAT), usually with a positive cut-off value at 1: 100 (Faine, 1982). The MAT test sensitivity is believed to be only moderate, hence diagnoses made at the individual-pig level are of limited value. For instance, in 20 sows that aborted and from which L. brutisluuu was isolated, only five reacted with ⩾ 1: 100 in the MAT test (Ellis et al., 1986b). This corresponds to a sow-level sensitivity at 25%. Thus the MAT test is primarily a herd test where a prevalence at 10% or more is indicated to be associated with an endemic infection (Ellis, 1992a). At the individual-pig level the use of paired samples to reveal a distinct rise in titer may be used. However, if two samples are taken l-4 weeks after clinical signs (abortion), paired samples may not be informative.
A positive serological result in a sow must be confirmed by direct visualization of the agent in suspect material by immunohistochemistry or by culture. On the other hand, the simple demonstration of antibodies in aborted fetuses strongly supports a definitive diagnosis (Bolin et al., 1991; Ellis, 1992b).
The prevalence of positive titers in sows from herds with reproductive problems is significantly higher than the prevalence in apparently normal herds (Hanson, 1988)) and associations exist between antibodies to L. brutisluua and infertility (Hathaway and Little, 1981). Further, split-herd clinical trials with a bacterin vaccine significantly improved the reproductive results in problem herds indicating that the speculated association between L. brutisluuu and a reproductive syndrome is real (Frantz et al., 1989).
In Denmark, L. brutisha has yet to be isolated, but seroreactions ⩾ 1: 100 in sows and boars have been detected in several studies (e.g. Nissen, 1989). In five of six herds with reproductive problems, the seroprevalence was 33%, and the incidence of animals with a rise in titer at an interval of 3-4 weeks was 13% (Mike1 Jensen and Binder, 1989). However, no apparent association between titer values and reproductive results was found, but this might have resulted from a lack of power given by the small sample size (93 sows). Persistence in the non-pregnant uterus is a special feature of L. brutisluuu (Ellis, 1992b), and recently, organisms indistinguishable from L. brutiskzuu were detected by immunofluorescence in non-pregnant sow uterus samples from a Danish herd with reproductive problems. Thus far, no large-scale serological investigation of L. brutisluuu infections has been conducted in Denmark. Accordingly, the present work was initiated in order to study: the distribution of L. brutisluuu MAT test titer values in Danish sow herds; the association between herd-level serological reactions and demographic data, herd-related factors, reproductive performance and incidence of medical treatments of sows; the age distribution of sows with positive titers.
Materials and methods
2.1. Selection of herds and sows
Forty-six herds, all volunteers, were selected in 1990 to participate in the Danish Health and Production Surveillance (HEPS) pilot project (Ellegaard et al., 1992; Christensen et al., 1994) ; 4 1 herds remained in the project until the summer of 1992. From this convenience sample, all herds with sows that had continued their usage of production data recordings were asked to participate in this study; 30 herds agreed to participate. All herds had computerized records available on production data, herd size and type, and on disease management system. No herds were vaccinated against any strain of leptospiras, including L. bratislaua. Some of the herds practice specific pathogen free (SPF) or minimal disease (MS) production. In the Danish model, SPF herds are free from Actinobacillus pleuropneumoniae (different serotypes), Mycoplasma hyopneumoniae, toxin producing strains of Pasteurella multocida, Serpulina hyodysenteriae, Sarcoptes scabiei and Haematopinus suis.
MS herds are equivalent to SPF herds except that they are infected with M. hyopneumoniae.
All sows and gilts (here defined as first-parity sows) were individually identified on the farms by an ear-tag as well as in the HEPS database by a sow-number. New gilts were assigned with individual identification at the latest at the date of their first service. For each farm, a list of all sows and gilts was prepared using the HEPS database. From this list, 20 sows and 15 gilts were selected at random (by use of a random number generator). All animals were selected irrespective of the reproductive cycles. Only animals that were not expected to be culled for the next 6 weeks (according to the farmer’s information) were included in the study. The animals were bled three times 6 weeks apart during the period from 26 January 1992 to 6 May 1992. At the first visit to the herds the farmers were questioned on their views on the herd’s reproductive performance including observations of symptoms that are usually characteristic for infections with L. bratislaua.
2.2. Serological analysis
Serum samples were analyzed by the MAT test for antibodies to the L. australis serogroup by using serovars L. bratislaua, strain Jez. The titers were expressed as at least 50% agglutination of leptospiras in dilutions 1:12.5, 1:25, 1:50, l:lOO, 1:200 and higher if necessary for end-point determination of the serum samples in isotonic NaCl solution. A cut-off value at 1: 100 was regarded as evidence of infection. Herds were categorized into three reaction groups: ‘ – ’ indicates no animal with a titer value > 1: 100; ‘ + ’ indicates one or more animal with a titer at > 1: 100 but no sows with an increase in titer by greater than two-fold between consecutive samplings; ‘ + + ’ indicates one or more animal with a titer > 1: 100 as well as one or more animal (which could be the same as the one with titer > 1: 100) with a > two-fold increase in titer between consecutive samplings. (Usually we consider a 2 two-fold increase to be an important seroconversion, and since in the data this was a relatively rare event (Table 2) a 2 two-fold increase was maintained as the criterion in the subsequent analysis.)
2.3. Preparation of datasets
Datasets for statistical analyses were obtained by merging information from the HEPS database with the results of the serological survey.
A herd-level dataset was prepared by calculating herd-level production parameter averages as well as incidence of medical treatments of sows and gilts for reproductive disorders. All results were obtained for a defined study period from 12 weeks prior to the date of the first blood sampling to 12 weeks after the last sampling (equal to a period from 20 October 1991 to 29 July 1992). Farrowing percentage was defined as the percentage that farrowed < 118 days after first service. Disease information included farmers’ recording of medical treatments given to sows and gilts in 28 of the 30 herds. The recordings of each treatment included date, animal-number and reason for treatment. The farmers did not attempt to make specific diagnoses but only recorded ‘respiratory disease’, ‘gastrointestinal disorder’, ‘lameness’, ‘disease at service’, ‘disease at farrowing’, ‘injury’, ‘systemic disease’ or ‘other disease’. Medical treatments given less than 6 days apart for the same reason were included as only one case. The recordings did not include routine treatments or vaccinations. From the HEPS database, the total number of sows at risk per farm per day in the study period was extracted (Christensen et al., 1994). Other herd characteristics, such as the number of sows and boars, and SPFIMS or conventional system, were added to the herd-level dataset. Finally, the geographical location of the herd was categorized into: Zealand and the island of Funen (postal district numbers 4000-5999); Jutland, south (postal district numbers 60006999); Jutland, middle part (postal district numbers 7000-8999); Jutland, north (postal district numbers 9000-9999).
Also an animal-level dataset was obtained including sows and gilts from the herds that had one or more seropositive animal. One observation was included for each sampled sow or gilt, defined as the farrowing cycles (weaning to weaning) in which the animal was serologically examined the most number of times, with a minimum of two serological results. Given the latter constraint, this dataset included 523 farrowings from 19 herds (in two herds, data on tested sows were only recorded for farrowing intervals in which the sow was tested only once).
2.4. Statistical analyses
In the herd-level analyses, demographic and herd-related factors were all categorized into two groups: a ‘risk’ group (as, for example, conventional herd) and a ‘non-risk’ group (as, for example, SPF/MS herd). Herd size and percentage boars (relative to the number of sows) were divided in two groups based on their median values: 141 sows and 3.97% boars, respectively. The influence of these factors for the risk of the herd falling into the combined category of ‘+’ or ‘+ +’ relative to the ‘-’ herds was evaluated by calculating crude odds ratios (OR) and by non-conditional maximum likelihood logistic regression (Kleinbaum et al., 1982). Two sets of models were evaluated. The first set included as explanatory variables: ‘conventional herd’ (relative to SPF/MS herd); ‘herd size at > 141 sows’ (relative to < 141); ‘> 3.97% boars’ (relative to < 3.97); ‘location outside northern Jutland’ (relative to location in this region). The second set included the same explanatory variables except the latter regional variable. A forward selection procedure was applied, employing a likelihood ratio test at each step, using SAS PROC LOGISTIC (Statistical Analysis Systems (SAS) Institute Inc., 1990). The significance level for entry into the model was set at 0.15. Since only a single main effect was found significant in either set, no interactions were subsequently tested.
In the analysis of herd-level reproductive performance, the continuous dependent variables (Table 6) were evaluated by multiple regression analysis for statistical association with the herd serological status, with control for herd size and average parity, using SAS PROC REG (SAS Institute Inc., 1988). Herd size was divided into two groups as described above. In all cases, the herd serological status (either ‘+ / + +’ combined or ‘-‘) was forced into the model. In a stepwise selection procedure, the significance of herd size and average parity was subsequently examined. The significance level for entry of the two variables into the model was set at 0.15.
In the description of serological reactions, prevalence was defined as the proportion of animals with titer values 3 1: 100. Cumulative incidence of serological reactors was defined as the proportion of sows with an increase in titer value between two consecutive samplings, and incidence density of medical treatments as the number of cases observed in the study period divided with the number of sow-days at risk (Kleinbaum et al., 1982). The herdlevel incidence densities for the three serological reaction types were compared using a non-parametric test (Kruskal-Wallis test; Armitage and Berry, 1987). In the animal-level analysis, two independent variables were defined. First, the material was divided into animals testing > 1:lOfl in any of the tests and animals with a constant lower titer. Second, the material was divided into animals with a greater than two-fold rise in titer and animals without this rise. The dependent variables included both continuous and categorical variables (Table 7). The continuous variables were evaluated by analysis of variance for statistical associations to the two independent variables including a random effect for herd, using SAS PROC MIXED (SAS Institute Inc., 1992). Parity as a fixed, categorical effect was subsequently tested, with a significance level for entry into the model at 0.15. The categorical dependent variables were tested for associations to the independent variables using a Fisher’s exact test. Because of low frequency of the categorical variables among animals with titers > 1: 100 or with a greater than two-fold rise in titer, no further modeling could be carried out.
Results
At the first visit to the farm, no pig producers reported any specific problems with reproductive failure. Since the HEPS database prior to the outset of the study was updated for each herd on a quarterly or bi-annual basis, some of the animals sampled in the database had been culled or were dead at the day of the first sampling. In addition, only animals that were not expected to be culled for the next 6 weeks were included in the study. Thus the original sample size of 1050 sows and gilts from the database was reduced to 807 actually tested
Table 1. Serological examinations of sows and gilts in 30 Danish swine herds in 1992 by the MAT test for antibodies against Leptospira bratislava: results of three consecutive samplings obtained 6 weeks apart

(B 1:lOO) were 22 (2.7%), 20 (2.5%) and 23 (2.9%). (If a cut-off of 150 was used instead, the prevalences were 9.3%, 11.4% and 9.9%, respectively.) The prevalence of reactors, when judged as positive in at least one of the three samples, is indicated in Table 2. Thus, serological reactions to L. bra&lava were not frequent at the individual-animal level, with only 4.5% reacting 2 1:lOO in one or more of the three repeated tests, and 2.2% with a two- or three-fold rise in titer.
Table 3 illustrates the herd-wise distribution of prevalences of titers > 1: 100 and the cumulative incidences of sows with an increase in titer. In only one herd was a two-fold increase in titer value observed without the presence of a titer value > 1: 100 in any of the three samples. This herd was assigned to the ‘-’ group. The prevalence of ‘+’ or ‘+ +’ herds was 21 (70%). Within the herds with positive sows, the seroprevalence of animals reacting > 1: 100 in one or more of the three tests ranged from 4% to 13%.
Serological reactions were detected in SPF/MS and conventional herds alike (Table 4), and the seroreactions were found in all geographical regions (yet relatively uncommon in northern Jutland).
In a bivariate analysis comparing ‘ + / + + ’ herds combined with the ‘ – ’ herds, only the geographical location (located outside northern Jutland compared to located within this region) and herd size > 141 sows were statistically related to the serological status (P= 0.003 and P = 0.054, respectively, using a Fisher’s exact test). In the logistic regression analysis including all four independent variables (health status with regard to SPF diseases, geographical location, herd size > 141 sows and boars as percentage of sows) only the geographical location was significant at the 15% level (OR = 19.0, P = 0.004)In. a similar analysis excluding the geographical location, a herd size > 141 sows was marginally significant (OR=5.41, P=O.O8).
Table 2. Serological examinations of sows and gilts in 30 Danish swine herds in 1992 by the MAT test for antibodies against Leptospira bratislava: prevalence in any of three samplings and cumulative incidence of serological reactors among 796 sows and gilts in three consecutive blood samplings, collected 6 weeks apart

Table 3. Serological examinations of sows and gilts in 30 Danish swine herds in 1992 by the MAT test for antibodies against Leptospira bratislava: prevalence of titers 2 1: 100 in sows and gilts in any of three consecutive samplings collected 6 weeks apart, and the frequency of sows with a rise in titers from one sampling to the next, in the 30 sow herds
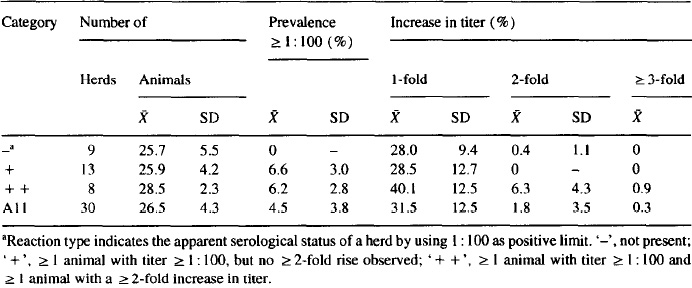
Table 4. Serological examinations of sows and gilts in 30 Danish swine herds in 1992 by the MAT test for antibodies against Leptospira bratisluva: relationship between serological results and the herd health status, the geographical location and the herd size
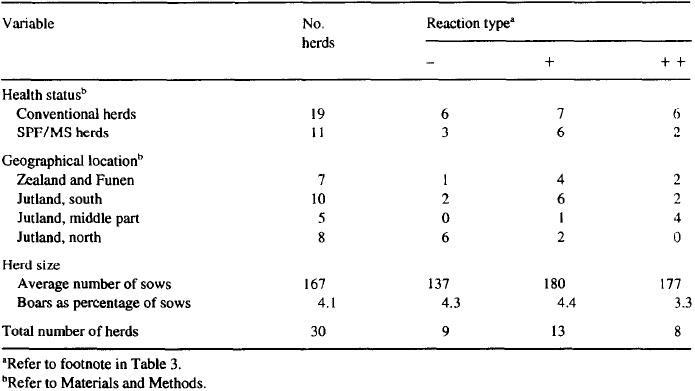
Table 5. Serological examinations of sows and gilts in 30 Danish swine herds by the MAT test for antibodies against Leptospira bratislava: regression analysis of production and reproductive results at the herd level
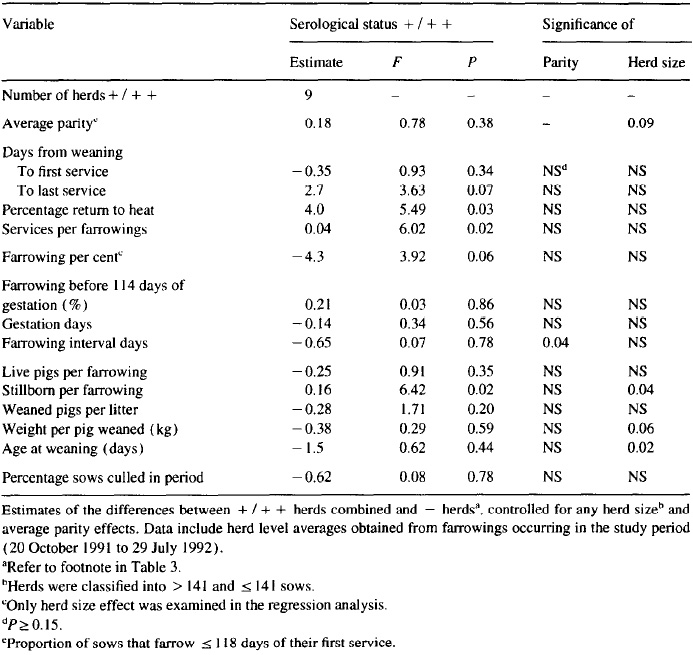
The results of the herd-level analysis on the association between positive titers and reproductive performance are presented in Table 5. Despite a low within-herd prevalence and cumulative incidence of positive sows, a statistical association between the serological status of the herd and important reproductive parameters could be inferred. In contrast to the production figures, no apparent detrimental effect of a positive serological status of the herd and incidence of treatments (Table 6) was found (total treatment incidence density P = 0.46; incidence density around service P = 0.33; incidence density around farrowing P = 0.24).
The distribution by parity of reactors >, 1: 100 and animals with increase and decrease in MAT titer values greater than two-fold is indicated in Fig. 1, for 523 farrowings originating from 19 positive herds. Only the difference in the prevalence of reactors 2 1:lOO was statistically significant (P = 0.05 in a Fisher’s exact test), while the cumulative incidences of animals with titer increase and decrease greater than two-fold were not different (P= 0.5 1 and P = 0.56, respectively).
Table 6. Serological examinations of sows and gilts in 30 Danish swine herds by the MAT test for antibodies against leprospira brarislava: mean and 10th to 90th percentile of medical treatment incidence density among sows and gilts in the study period (20 October 1991 to 29 July 1992)
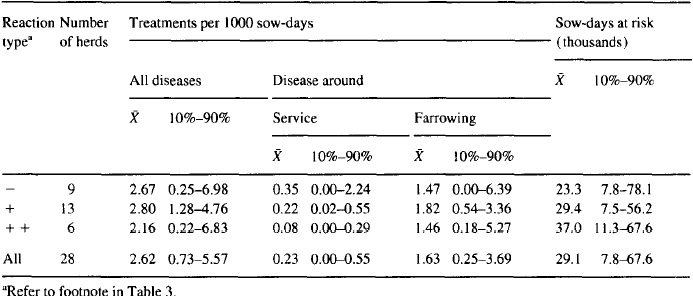
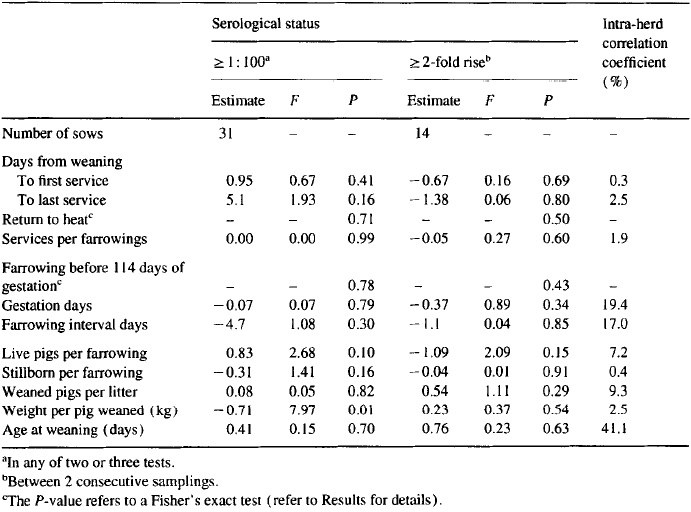
Analysis of the reproductive performance at the individual-animal level revealed no apparent association between the reproductive performance of the animal and its serological status ( > 1: 100 in any test, or greater than two-fold increase in titer; Table 7). Parity as a fixed effect was found significant (P < 0.15) concerning the variables ‘days from weaning to first service’ (P = 0.004)) ‘days from weaning to last service’ (P = 0. lo), ‘services per farrowing’ (P = 0.11)) ‘live pigs per farrowing’ (P < 0.001)) ‘weaned pigs per litter’ (P = 0.002)) and ‘age at weaning’ (P = 0.10). For animals with titer values > I: 100, the frequencies of return to heat and farrowings before 114 days of gestation were 4 ( 12.9%) and 1 (3.23%), respectively. For animals with a rise greater than two-fold in titer, the similar frequencies were 1 (7.1%) and 0 (0.0%)) respectively.

Fig 1. Serological examinations of sows and gilts in 19 Danish swine herds in 1992 testing positive by the MAT test for antibodies against Leprospira brarislaoa. The distribution of serological MAT reactors by parity. Results from 523 sows and gilts.
Table 7. Serological examinations of 523 sows and gilts in 19 Danish swine herds in 1992 testing positive by the MAT test for antibodies against Leptospira bratisfava: estimates of the differences between animals with 2 1: 100 in any test and animals with a constant lower titer, and of the differences between animals with a 2 2-fold rise in titer and animals without this rise (results are controlled for parity and intra-herd correlation effects)
DISCUSSION
The selection of herds for the study should be considered. All 30 examined herds participated in a continuing evaluation of production diseases (Christensen et al., 1994), and no pig producer reported a history of recurrent reproductive problems that usually are characteristic for infections with L. brutisZuua. The farmers’ opinion was supported by the fact that the prevalence of sows that returned to heat in the studied herds (6.9%) was below the current Danish average of 7.4% (Helm, 1993).
Also, the external validity of the sample of herds needs consideration. The average size of the 30 herds, 167 sows per herd, is considerably larger than the Danish average (currently at approximately 50 sows per herd), and results from the study indicated that a herd size effect may be significant (Table 4). The frequency of SPF/MS herds in the sample is 11 out of 30 (37%), far above the national average (approximately 9%)) but this factor did not appear to be associated with positive serological findings. Thus, in our view, the results of the survey estimate the prevalence and incidence of seroreactions to L. bratislam in (apparently) normal, relatively large-sized Danish sow herds, irrespective of their health status regarding SPF diseases.
The herd-level analysis was carried out on only 30 observations, hence caution should be exercised when evaluating the results. The analysis indicated that the herds’ disease status (SPF/MS or conventional) was of little importance. The SPF/MS system provides no control for L. bratislava infections, and the more stringent quarantine and disinfection procedures practised in such herds apparently have little if any effect on the risk of infections. Similar results were found concerning the relative number of boars (all herds in the study were using artificial insemination as well as boars). This might indicate that the number of boars needed to maintain an infection with L. bratidaoa is relatively low. The apparent positive association between herd size and the risk of a herd being seropositive is,not unexpected, since most epidemiological studies on productive and infectious diseases in the Danish swine population have indicated this relationship (Gardner et al., 1995). Finally, the regional effect, with only few serologically positive herds in northern Jutland, may either be biological or be caused by herd-size confounding. This in our view cannot be evaluated in the present study, given the relatively low sample size of herds.
The prevalence and cumulative incidence of seroreactors (Tables 1 and 2) were considerably lower than in the previously reported Danish study on six herds with reproductive problems (Mike1 Jensen and Binder, 1989). However, an association was detected between a herd’s serological status and its reproductive performance, with focus on the prevalence of sows returning to heat and the number of stillborn pigs (Table 5). Thus, the results are in concordance with previous studies, where it has been demonstrated that L. bratislaua mainly causes an infertility syndrome (Hathaway and Little, 198 1; Ellis, 1992b), associated with a characteristic tendency of persistence of L. brutisha in the oviduct and uterus (Ellis et al., 1986b). It has also been established that chronically infected animals often do not possess MAT titer values at or above the accepted cut-off of 1: 100 (Ellis, 1992b). In conclusion, the findings are not contradictory to the results of previous research that demonstrates a direct or indirect association between L. bratislava and reproductive failure (Frantz et al., 1989; Bolin et al., 1991).
However, the results of the study only yield circumstantial evidence of causality between L. bratislava and infertility in Danish sow herds. First, no association between the herd serological status and the incidence density of treatments could be detected. Second, the cross-sectional study design does not allow for an evaluation of the timing between infection and disease event. Third, the agent itself has yet to be isolated from Danish herds. The actual presence of L. bratislaua in Denmark has been confirmed only by immunofluorescence in material from a problem herd not related to the present study. In consequence, epidemiological cohort studies, and an intensified effort for direct or indirect demonstration of the agent, are both recommended. The apparent lack of (systematic) associations between serological status and reproductive performance at the animal level was a feature of this study, and this has previously been reported elsewhere (Mike1 Jensen and Binder, 1989; De Waal et al., 1991). However, such findings are not unexpected, given the low sensitivity of the MAT test. Probably, a large proportion of seronegative sows are incorrectly classified as non-infected. A marked pattern of serological response by parity was observed (Fig. 1) . Apparently, the primary exposure to L. bratislaoa takes place early, given the high prevalence of positive reactions but low cumulative incidence of gilts with titer increase at the first parity. In firstlitter gilts, the first blood sample was obtained (on the average) 10 weeks after the animal was introduced into the herd (typically defined as the date of the first service), and the second sample 6 weeks later. Thus, seroconversion must have taken place prior to 16 weeks after the first service. Then immunity, either at the level of the individual sow or at the herd level, gradually declines, with low prevalence and cumulative incidence of titer increase in the second and third parity. This allows for a second peak at the fifth parity, with a high prevalence, increased incidence of titer increases and no incidence of titer decreases. It should be stressed, however, that the observed within-herd serological status by age may be confounded by the cross-sectional design of the study.
References
Armitage, P. and Berry, G., 1987. Statistical Methods in Medical Research. Blackwell Scientific, London, pp. 93- I40,37 I-420.
Baker, T.F.. McEwen, S.A.. Prescott, J.F., Meek, A.H. and Waltertoews, D., 1988. The prevalence of leptospirosis and its association with multifocal interstitial nephritisin swineat slaughter. Proceedingsofthe 5th International Symposium on Veterinary Epidemiology and Economics. Acta Vet. Stand. Suppl., 84: 306308.
Bercovich, Z., Spek, C.W. and Comvalius-Adrin, I., 1983. Occurrence of antibodies to various serotypes of Leprospiro interrogum among swine in the Netherlands between 1975 and 1980. Tidjschr. Diergeneeskd., 108: 133-138.
Bolin. C.A. and Cassells. J.A., 1990. isolation of Lepfospiru interroguns serovar bratislmw from stillborn and weak pigs in Iowa. J. Am. Vet. Med. Assoc., 196: 1601-1604.
Bolin, CA., Cassells, J.A., Hill. H.T., Frantz, J.C. and Nielsen, J.N.. 1991. Reproductive failure associated with Lepto.spiru interrogunv serovar bro/i.hru infection of swine. J. Vet. Diagn. Invest., 3: 152-154.
Christensen, J., Ellegaard, B., Kirkegaard Petersen, B., Willeberg, P. and Mousing, J., 1994. Pig health and production surveillance in Denmark. Sampling design, data recording and measures of disease frequency. Prev. Vet. Med., 20: 47-61.
De Waal. C.A.H., Hartman, E.G., Bokhout, B.A. and van Leengoed, L.A.G.M., 1991. Porcine leptospirosis. Remarks on the sero-diagnosis of Leprospiro interrogans serotype brurishw. Tijdschr. Diergeneeskd., 116: 173-179.
Egan, J. and Yearsley, D., 1987. Serological survey of leptospiral antibodies in cattle, sheep and pigs in the Republic of Ireland. lr. Vet. J., 46: 213-214.
Ellegaard, B., Herlgv, L., Tybirk, P., Christensen, J., Kirkegaard Petersen, B. and Mousing, J., 1992. Bes=tningsSundhedsOvervSgning (BSO) i svinebesaetninger. Dan. Vet. Tidsskr., 75: 269-275.
Ellis, W.A., 1992a. Leptospirosis. In: A.D. Leman, B.E. Straw, W.L. Mengeling, S. D’Allaire and D.J. Taylor (Editors), Diseases of Swine, 7th edn. Iowa State University Press, Ames, pp. 529-536.
Ellis, W.A., 1992b. Leptospirosis in pigs. Pig Vet. J., 28: 24-34.
Ellis, W.A. and Thiermann, A.B., 1986. Isolation of Leprospiru inrerrogans serovar brotislova from swine in Iowa. Am. J. Vet. Res., 47: 1458-1460.
Ellis, W.A., McParland, P.J., Bryson, D.G. and Cassells, J.A., 1986a. Boars as carriers of leptospiras of the Australis serogroup on farms with an abortion problem. Vet. Rec., 118: 563.
Ellis. W.A., McParland, P.J., Bryson, P.J., Thiermann, A.G. and Montgomery, J., 1986b. Isolation of leptospiras from genital tract and kidneys of aborted sows. Vet. Rec., 118: 294-295.
Faine, S., 1982. Guidelines for the Control of Leptospirosis. World Health Organization, Geneva. Frantz, J.C., Hansson, L.E. and Brown, A.L., 1989. Effect of vaccination with a bacterin containing Leptospira inferrogans serovar brutiskzva on the breeding performance of swine herds. Am. J. Vet. Res., 50: 1044-1047.
Gardner, 1.. Willeberg, P. and Mousing, J., 1995. Herd size as a risk factor for disease in swine farms. Prev. Vet. Med., in press. Hanson, L.E., 1988. Leptospira bratislaua infection: serologic incidence. In: Proceedings: Symposium on Leptospira hrafish~a infection, 13 December 1988, Lincoln, Nebraska, pp. 9-1 I.
Hathaway, S.C. and Little, T.W.A., 1981. Prevalence and clinical significance of leptospiral antibodies in pigs in England. Vet. Rec., 108: 224-228.
Holm, M., 1993. Report on production results from Danish swine herds 1992-1993. Danish Farm Agricultural Center, Skejby, Denmark, unpublished. Kleinbaum, D.G., Kupper, L.L. and Morgenstem, H., 1982. Epidemiologic Research. Principles and Quantitative Methods. Van Nostrand Reinhold, pp. 140-158,419-l46.
Mike1 Jensen, A. and Binder, M., 1989. Seroteaktion for leptospirose og reproduktionsproblemer hos danske svin. Er der en sammenhaeng? En forelobig undersogelse. Dan. Vet. Tidsskr., 72: 1181-I 186.
Nissen, O.D., 1989. Porcin leptospirose. En litteraturoversigt samt tidsforlb for L. bratis1ar.a i to besaztninger. Dan. Vet. Tidsskr., 46: 619-634. Sandstedt. K. and Engvall, A., 1985. Serum antibodies to Leptospira bratislalfa in Swedish pigs and horses. Nord. Veterinaermed., 37: 3 12-3 13.
Statistical Analysis Systems Institute Inc., 1988. SAS/STAT User’s Guide, Release 6.03 Edition, Gary, NC. Statistical Analysis Systems Institute Inc., 1990. Technical Report P-200, SAS/STAT Software: CALIS and LOGISTIC Procedures, Release 6.04, Statistical Analysis Systems Institute Inc., Gary, NC. Statistical Analysis Systems Institute Inc., 1992. SAS Technical Report P-229, SAS/STAT
Software: Changes and Enhancements, Release 6.07. Statistical Analysis Systems Institute Inc., Gary, NC. Schonberg, A., Barbel, H.-H., Klmpe, U., Schmidt, K. and Ellis, W.A., 1992. The isolation and identification of Leptospira interrogans serovar bratisha from a pig in Germany. J. Vet. Med. B, 39: 362-368. Smith, K.E., Zimmerman, J.J., Bolin, CA., Beran, G.W. and Hill, H.T., 1992. A survey of house mice from Iowa swine farms for infection with Leptospiru inferrogans serovar hratisha. Can. Vet. J., 33: 742-744.

